Encapsulation
- Post by: Leo
- January 6, 2021
- Comments off
Encapsulation: Bacteria, yeast, cell, surfactant, vitamin, drug, colorant, flavor, and fragrance
Works have been accomplished…
-
Enhance the heat stability of cinnamon flavor by ultrasonication-assisted microencapsulation and reduce the interaction of encapsulated cinnamaldehyde with yeast for baking applications
-
Encapsulate the lipophilic bioactive components (e.g., fish oil) to improve the oxidative stability and shelflife
-
Improve the solubility and bioavailability of drugs (e.g., doxorubicin) for targeted delivery
-
Develop aqueous core−shell−shell coconut-like nanostructure for stimuli-responsive delivery of hydrophilic cargo
-
Design a core−shell-structured microcapsule to release the cargo only when exposed to lipase
-
Develop a microfluidic platform for encapsulation of enzymes within microcapsules for controlled release
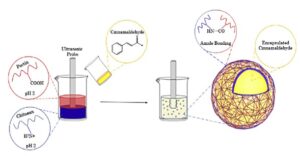
Cinnamaldehyde encapsulation schematic detailing capsule formation via ultrasonication-induced polymeric cross-linking of pectin and chitosan polysaccharides.
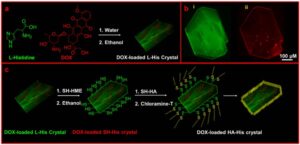
(a) Schematic representation for the preparation of L-His crystals loaded with DOX molecules. (b) CLSM images of (i) L-His crystal emitting green color, (ii) DOX with red color in the L-His crystal. (c) Schematic representation for the preparation of L-His crystals surfacemodified with tumor-specific HA for the targeted delivery of hydrophobic DOX molecules.
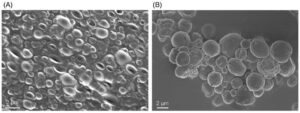
SEM images of microcapsules corroborating capsule formation and morphology: (A) CA-UCM and (B) SD-CA-UCM.
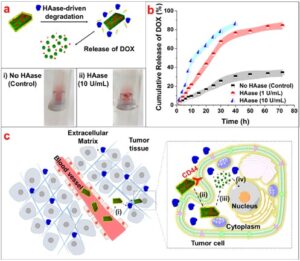
Enzymatic degradation of HA-His crystals in the presence of 1 or 10 U/mL HAase at 37 °C. (a) Schematic of HA-His crystals that can be degraded by HAase, and digital images of the HA-His crystals after 4 h without the presence of HAase [control, (i)] and in the presence of Haase (ii). (b) Cumulative release of DOX from HA-His crystals. (c) Schematic of the enhanced delivery of hydrophobic chemotherapeutics by the HAHis crystals for cancer therapy: (i) HA-His crystals accumulate in the tumor; (ii) HA-His crystals are internalized by the CD44 receptors on the tumor cells; (iii) HAase leads to the degradation of HA on the crystal surface, dissolving the crystals; and (iv) release of the hydrophobic chemotherapeutics over time to cause the tumor cell death.
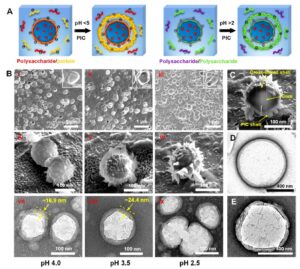
An additional PIC shell generated on the surface of the shell cross-linked nanocapsules. (A) Schematic illustrating the electrostatic deposition of non-cross-linked polyelectrolytes back onto the surface of the cross-linked shell after the pH was switched. The combinations of polysaccharide/protein and polysaccharide/polysaccharide were taken as an example. (B) Morphological evolution of the nanocapsules with decreasing pH from 5.0 to (i, iv, vii) 4.0, (ii, v, viii) 3.5, and (iii, vi, ix) 2.5. (i−iii) SEM, (iv−vi) cryo-SEM, and (vii-ix) TEM images, respectively. (C) A cross-sectional image produced by cryo-FIB milling, showing high magnification of the core−shell−shell nanostructure. Here, the capsules were made by decreasing the pH from 5.0 to 2.5, with CHS and BSA as the shell materials, that is, ATC-CHS-BSA. (D, E) TEM images of fully polysaccharide capsules made of (D) pectin-chitosan and (E) xanthan gum-chitosan formed by increasing the pH from 2.0 to 4.6.
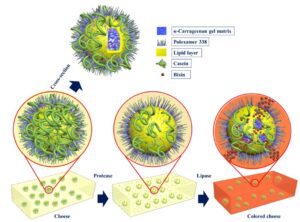
Schematic of the enzymatically triggered microcapsules and their controlled release by lipase.
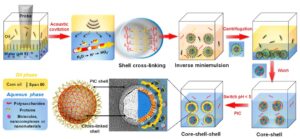
General strategy for the synthesis of an aqueous core−shell−shell nanostructured platform via sequential cross-linking and PIC. Here, the combination of polysaccharides and proteins is taken as an example to fabricate a cross-linked nanocapsule shell, followed by the direct deposition of excess non-cross-linked proteins to form an additional PIC shell layer.
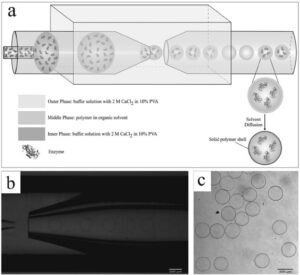
a) Schematic illustration of encapsulation of enzymes using microfluidics. b) Optical image showing production of microcapsules at the tip of the injection capillary. c) Optical image of as-prepared microcapsules.

(a) Microscopy images of the microcapsules taken using light microscopy and (b, c) SEM images of microcapsules; (d−f) cryo-SEM of microcapsules cross-sectioned by focused ion beam.
Peer-reviewed Publications
-
Ultrasonic encapsulation of cinnamon flavor to impart heat stability for baking applications (https://doi.org/10.1016/j.foodhyd.2019.105316)
-
L-Histidine Crystals as Efficient Vehicles to Deliver Hydrophobic Molecules (https://doi.org/10.1021/acsami.9b14239)
-
A Robust Aqueous Core–Shell–Shell Coconut-like Nanostructure for Stimuli-Responsive Delivery of Hydrophilic Cargo (https://doi.org/10.1021/acsnano.9b03049)
-
Combination of internal structuring and external coating in an oleogel-based delivery system for fish oil stabilization (https://doi.org/10.1016/j.foodchem.2018.10.112)
-
Controlling the release from enzyme-responsive microcapsules with a smart natural shell (10.1021/acsami.7b18795)
-
Osmotic pressure triggered rapid release of encapsulated enzymes with enhanced activity (https://doi.org/10.1002/adfm.201700975)
Categories: